Oncocytic Variant of Adrenocortical Carcinoma: Case Report of a Rare Malignancy
A B S T R A C T
Adrenocortical carcinoma is a rare cancer. Oncocytic tumors of the adrenal gland are rarer. Most Oncocytic Adrenal Neoplasms are benign and carry favourable prognosis. They are classified as oncocytoma, oncocytic neoplasm of uncertain malignant potential and oncocytic adrenal carcinoma. The malignant nature of oncocytic neoplasm of adrenal gland can only be confirmed on histopathology. We report a case of a 55-year-old male with newly diagnosed hypertension being evaluated for left adrenal mass concerning for adrenocortical carcinoma. Open radical left adrenalectomy and nephrectomy was done and histopathology confirmed oncocytic variant of adrenocortical carcinoma based on Lin-Weiss-Bisceglia scoring system which has been developed particularly for oncocytic type of tumor. Though rare, oncocytic neoplasm has to be considered as one of the differential diagnoses of adrenocortical mass, especially those presenting as a large mass because malignant oncocytic neoplasm of adrenal gland as large as 23cm have been reported. Imaging modalities like ultrasonography, computed tomography or magnetic resonance imaging, though useful in evaluating an adrenocortical mass, cannot predict malignant nature of an oncocytic neoplasm. Diagnosis of adrenocortical carcinoma is therefore reliably made only after histopathological examination of the surgical specimen. Surgical resection in those presenting with non-metastatic resectable disease remains the mainstay of ACC treatment. Oncocytic ACC compared with conventional ACCs matched for age, gender, disease stage and status of surgical resection, shows significant better overall survival thus representing more indolent variant of an aggressive and often fatal disease.
Keywords
Adrenal tumor, oncocytic adrenal neoplasm, adrenocortical carcinoma, oncocytic adrenocortical carcinoma, adrenalectomy
Introduction
Adrenocortical carcinoma (ACC) is a rare primary adrenal neoplasm with an incidence of 1-2 cases per million per year [1]. Oncocytic variant is a rarer subtype that represent approximately 10% of adrenocortical tumors [2]. We report a case of oncocytic ACC with emphasis on management, histopathological features and immunohistochemical profile of the tumor aiding in its distinction from the benign adrenocortical oncocytoma and conventional ACC.
Case Presentation
A 55-year-old man was referred to our hospital for management of a left suprarenal mass detected on ultrasonography done for evaluation of persistent pain in his left hypochondrium. He was recently diagnosed to have hypertension and diabetes mellitus. He did not have episodic headache/ palpitation/ diaphoresis. His blood pressure at presentation was 150/90 mmHg. He had no physical features suggestive of adrenocortical hyperfunction. Examination of the abdomen revealed a mass occupying the left hypochondrium and the left lumbar region.
His serum cortisol, metanephrine and normetanephrine levels were within normal limits while dehydroepiandrosterone (DHEA) was elevated. His renal and liver function tests were normal. Contrast enhanced CT scan of the chest and abdomen revealed an 8.5X9X13.5 cm left adrenal mass. The lesion had areas of low attenuation in the centre, suggestive of necrosis. Heterogeneous contrast enhancement was noted in the arterial phase. The lesion was infiltrating superior pole of the left kidney. Open adrenalectomy was planned. Intraoperatively there was a 10X8X12cm left adrenal lesion infiltrating the left kidney. Multiple enlarged nodes were present. Radical adrenalectomy and nephrectomy were performed. He had an uneventful postoperative recovery.
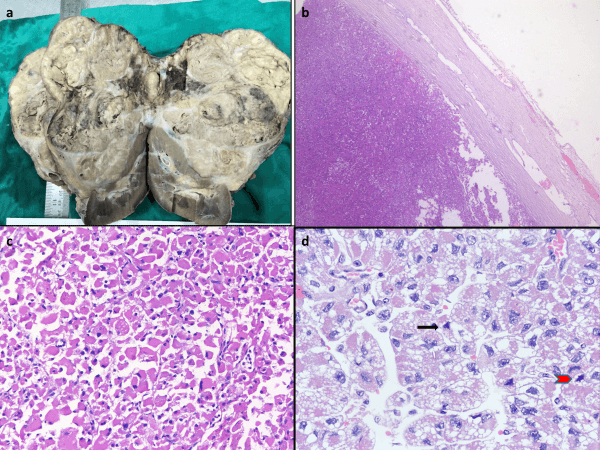
Figure 1: a) Large tumor pushing the kidney, on cut surface showing grey white with necrosis, b) Tumor with the capsule(H&EX40), c) Oncocytic cells with abundant eosinophilic cytoplasm(H&EX200) d) Increased mitotic activity in the tumor, the arrow shows an atypical mitosis(H&EX400).
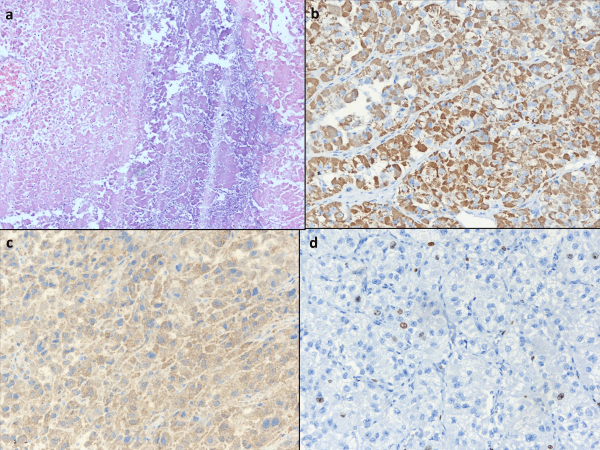
Figure 2: a) Showing necrosis (H&EX100), b) Tumor cells show strong immunohistochemistry positivity for Melan A, c) for calretinin, d) Ki67 showed proliferative index of 8%.
Histopathological examination revealed encapsulated tumor with cells arranged in nests, cords and sheets with abundant eosinophilic to clear cytoplasm, eccentrically placed nuclei with coarse chromatin (Figure 1). Clear cells comprised less than 25% of tumor cells. Extensive necrosis and haemorrhage were present. The mitotic count was 5/10 high power field (HPF) (Figure 1). The Modified Weiss score was 6. The Immunohistochemical analysis revealed that the tumor cells were positive for melan A and negative for inhibin, CD117, and chromogranin (Figure 2).
Discussion
I Introduction and Epidemiology
The term “Oncocytoma” was first used by Hamperl to describe a tumor composed of large cells with highly granular eosinophilic cytoplasm owing to the high mitochondrial content in the cytoplasm. Oncocytic neoplasms are commonly described in thyroid, parathyroid, salivary glands, and kidney and few in respiratory tract and choroid plexus [3]. The oncocytic adrenal neoplasms (OAN) are rare variant with only few cases described so far in the literature. Being rare, most of the available data are from retrospective case reports and case series and prospective studies investigating potential therapies are limited. Approximately 20% of OAN show some features of malignancy and 30% of them exhibit hormone production [4]. They are more common in women and tend to affect the left side more frequently.
II Clinical Presentation
OANs are usually large tumors at presentation. However, size of the mass and imaging findings (abdominal CT scan and MRI) are not pathognomonic of malignant characteristics [5]. Very rarely they present with features of hormone hypersecretion like Cushing’s syndrome, hyperaldosteronism, and virilization [6].
III Establishing Diagnosis
Contrast enhanced CT scan and MRI are used for assessment of adrenal incidentalomas. Although imaging characteristics can help to predict the malignant nature, no specific features are described for OAN [6]. There are few published reports about (18F)-FDG whole-body positron emission tomography findings in OAN with contradictory results [6]. Even FNAC and percutaneous biopsy are not adequate to distinguish OAN from other adrenal lesions or establish its malignant potential [6]. Therefore, it is not possible to accurately diagnose or predict oncocytic neoplasm or its malignant potential during preoperative evaluation. Endocrine evaluation preoperatively is mandated in patients with adrenal tumors. OACs may produce hormones more frequently than reported in literature [3]. Malignant adrenal masses secrete a higher ratio of DHEA than benign adenomas. Other tests include aldosterone-to-renin ratio, testosterone, estrone, 17β-estradiol, 17-OHprogesterone, androstenedione, and 24-hour urinary 17-corticosteroids measurements [1]. This patient had an elevated DHEA with no clinical evidence of hormonal imbalance.
IV Postoperative Histopathology and Immunohistochemistry (IHC)
Histopathological examination shows oncocytes which are 1-2 times the normal adrenocytes with abundant eosinophilic granular cytoplasm due to accumulation of mitochondria (almost 60 % of the cytoplasm), central pyknotic nucleus and a prominent nucleoli [3, 6]. Weiss scoring system was most widely used in the past [7]. Diagnosis of ACC required three or more of the nine criteria to be met. However, Weiss score overestimates the malignancy due to intrinsic characteristics of the oncocytic cells i.e., eosinophilic, elevated Fuhrman(nuclear) grade, diffuse architectural structure making a minimum score of 3 for oncocytic neoplasm. Thus, alternative scores have been proposed [2]. Biscelgia et al. revised Weiss criteria and proposed the Lin–Weiss–Bisceglia score which is currently recommended for OAN: major criteria- mitotic rate > 5 mitoses per 50 high power fields, any atypical mitoses or venous invasion; minor criteria- > 10cm or > 200 g, necrosis, capsular invasion or sinusoidal invasion; diagnostic criteria for OAN- malignant (OAC): any major criteria, borderline oncocytic neoplasm of uncertain malignant potential: only minor criteria and benign (adrenocortical oncocytoma): no major, no minor criteria [2, 7].
Our patient fulfilled both the major criteria, i.e. mitotic rate > 5 mitoses per 50 HPF and atypical mitoses indicating malignant variant of oncocytic neoplasm. The Helsinki score which has been recently proposed incorporates the Ki-67 proliferation index and is considered a representative prognostic score for OAC [2].
When dealing with an oncocytic tumor, its adrenocortical nature is confirmed by IHC to avoid misdiagnosis as pheochromocytoma. IHC profile of primary adrenocortical lesions shows variable positivity with calretinin, melan-A, alpha- inhibin and synaptophysin and are chromogranin negative. Adrenal pheochromocytomas are 100% positive for synaptophysin and chromogranin and negative for calretinin, inhibin and melan-A distinguishing these tumors from adrenocortical lesion [8].
Oncocytic variants of ACC have cells with eosinophilic granular cytoplasm which are diffusely and strongly immunostained with antimitochondrial antibody due to increase in the number of mitochondria supporting the recognition of OAC [7, 9]. This patient showed positivity only to melan A and negative for inhibin, CD 117, chromogranin and Ki67 proliferative index of 8%.
V Treatment Options
Tumor growth is the major cause of death in patients with ACC. At the time of diagnosis, based on imaging, the disease is classified according to European Network for the Study of Adrenal Tumors as stage I, II, III, or IV [10]. In our centre, FDG-PET/CT is not routinely performed at initial staging. Complete surgical removal (open/laparoscopic/robotic) of the tumor remains the only curative treatment option for patients with ACC. However, even after R0 resection the cumulative recurrence rate is high ranging 30–75% [11]. As opposed to earlier concepts contraindicating (relative) laparoscopic approach for larger tumors (>8cm), laparoscopic lateral transabdominal adrenalectomy for a large (120×95×110 mm) oncocytic ACC has been reported establishing laparoscopy as a safe and effective approach in experienced hands [6, 12].
Patients with locally advanced disease require en-bloc resection of adjacent organs and routine nephrectomy is not recommended in all patients. Locoregional lymph node dissection may improve tumor staging and have a favourable oncologic outcome in patients with localized ACC but its extent remains unspecified [13]. This patient underwent radical open left adrenalectomy and nephrectomy. Mitotane is the only drug approved (by the European Medicines Agency and the FDA) for the treatment of patients with advanced and inoperable ACC [14]. Neoadjuvant treatment with mitotane or other compounds (etoposide, doxorubicin, and cisplatin) as a part of treatment protocol are yet to be studied [3]. Recommendations on adjuvant therapy with mitotane, cytotoxic chemotherapy, and radiation therapy even for conventional ACCs are based on largely retrospective, limited data [3]. Guidelines for adjuvant treatment of OACs generally follows that of conventional ACCs; however, because oncocytic variants have a more favourable biologic behaviour, this extrapolation may impact the expected therapeutic benefits. Hence further studies on OACs are warranted.
VI Prognosis
The prognosis of overall ACC is more heterogeneous. OACs follow an indolent course, have delayed recurrence and improved overall survival [2, 3].
VII Follow Up Guideline
No specific guidelines pertaining to OAC have been suggested. After surgery, adrenal oncocytic benign neoplasm are assessed for 5 years. OAC is more aggressive and may present with local invasion or with metastasis. 5-year survival after surgery is 50-60 %. Overall median survival for OAC is 58 months suggesting prognosis of malignant OANs is more favourable than conventional ACCs (median survival 14-32 months) [4].
Conclusion
Oncocytic variant of ACC is a rare variant that tend to be of lower stage, less locally advanced and have improved survival with fewer recurrences compared to conventional ACCs. Awareness of this OAC subtype of ACCs may help guide future treatment and result in more accurate predictions for patient prognosis. Resection is the mainstay of treatment with curative intent. Benefits of postoperative systemic therapy and adjuvant radiotherapy, more so in patients with greater risk for recurrence are still investigational in cases of oncocytic ACC. Previous studies include only small cohorts of patients owing to its low prevalence. Hence, further prospective studies with a joint multi-institutional effort is needed before definitive recommendations can be made in this regard.
Abbreviations
ACC: Adrenocortical Carcinoma
DHEA: Dehydroepiandrosterone
HPF: High Power Field
OAN: Oncocytic Adrenal Neoplasms
Acknowledgement
None.
Conflicts of Interest
None.
Consent
Consent for publication was taken from the patient.
Article Info
Article Type
Case ReportPublication history
Received: Tue 12, May 2020Accepted: Fri 29, May 2020
Published: Mon 08, Jun 2020
Copyright
© 2023 Sarath Sistla. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.SCR.2020.06.06
Author Info
Balamourougan Krishnaraj Gomathi Shankar Jigish Ruparelia Prakriti Giri Sarath Sistla Srinivas Bheemanathi Hanuman
Corresponding Author
Sarath SistlaDepartment of Surgery, Jawaharlal Institute of Post-Graduate Medical Education and Research, Puducherry, India
Figures & Tables
References
- Wong KK, Miller BS, Viglianti BL, Dwamena BA, Gauger PG et al. (2016) Molecular Imaging in the Management of Adrenocortical Cancer: A Systematic Review. Clin Nucl Med 41: e368-e382. [Crossref]
- Renaudin K, Smati S, Wargny M, Al Ghuzlan A, Aubert S et al. (2018) Clinicopathological Description of 43 Oncocytic Adrenocortical Tumors: Importance of Ki-67 in Histoprognostic Evaluation. Mod Pathol 31: 1708-1716. [Crossref]
- Mills JK, Khalil M, Pasieka J, Kong S, Xu Y et al. (2019) Oncocytic Subtypes of Adrenal Cortical Carcinoma: Aggressive in Appearance Yet More Indolent in Behavior? Surgery 166: 524-533. [Crossref]
- Wong DD, Spagnolo DV, Bisceglia M, Havlat M, McCallum D et al. (2011) Oncocytic Adrenocortical Neoplasms--A Clinicopathologic Study of 13 New Cases Emphasizing the Importance of Their Recognition. Hum Pathol 42: 489-499. [Crossref]
- Corrales JJ, Robles Lázaro C, Sánchez Marcos AI, González Sánchez MC, Antúnez Plaza P et al. (2016) Plurihormonal Cosecretion by a Case of Adrenocortical Oncocytic Neoplasm. Case Rep Endocrinol 2016: 6785925. [Crossref]
- Mearini L, Del Sordo R, Costantini E, Nunzi E, Porena M (2013) Adrenal Oncocytic Neoplasm: A Systematic Review. Urol Int 91: 125-133. [Crossref]
- de Krijger RR, Papathomas TG (2012) Adrenocortical Neoplasia: Evolving Concepts in Tumorigenesis With an Emphasis on Adrenal Cortical Carcinoma Variants. Virchows Arch 460: 9-18. [Crossref]
- Sangoi AR, McKenney JK (2010) A Tissue Microarray-Based Comparative Analysis of Novel and Traditional Immunohistochemical Markers in the Distinction Between Adrenal Cortical Lesions and Pheochromocytoma. Am J Surg Pathol 34: 423-432. [Crossref]
- Ohtake H, Kawamura H, Matsuzaki M, Yokoyama E, Kitajima M et al. (2010) Oncocytic Adrenocortical Carcinoma. Ann Diagn Pathol 14: 204-208. [Crossref]
- Fassnacht M, Johanssen S, Quinkler M, Bucsky P, Willenberg HS et al. (2009) Limited Prognostic Value of the 2004 International Union Against Cancer Staging Classification for Adrenocortical Carcinoma: Proposal for a Revised TNM Classification. Cancer 115: 243-250. [Crossref]
- Fassnacht M, Johanssen S, Fenske W, Weismann D, Agha A et al. (2010) Improved Survival in Patients With Stage II Adrenocortical Carcinoma Followed Up Prospectively by Specialized Centers. J Clin Endocrinol Metab 95: 4925-4932. [Crossref]
- Panizzo V, Rubino B, Piozzi GN, Ubiali P, Morandi A et al. (2018) Laparoscopic Trans-Abdominal Right Adrenalectomy for a Large Primitive Adrenal Oncocytic Carcinoma: A Case Report and Review of Literature. Am J Case Rep 19: 1096-1102. [Crossref]
- Reibetanz J, Jurowich C, Erdogan I, Nies C, Rayes N et al. (2012) Impact of Lymphadenectomy on the Oncologic Outcome of Patients With Adrenocortical Carcinoma. Ann Surg 255: 363-369. [Crossref]
- Crona J, Beuschlein F (2019) Adrenocortical Carcinoma - Towards Genomics Guided Clinical Care. Nat Rev Endocrinol 15: 548-560. [Crossref]
