Journals
Procedure and Effectiveness of Extracorporeal Membrane Oxygenation in Percutaneous Coronary Intervention for High-risk Complex Coronary Disease
A B S T R A C T
Objective: It has been demonstrated that performing percutaneous coronary intervention (PCI) in the absence of mechanical circulation support (MCS) for patients with complex high-risk coronary artery disease bears a high risk. Alternatively, to figure out the procedure effectiveness and the mid-term prognosis of PCI for complex high-risk coronary artery disease, we accomplished the whole process by the assistance of extracorporeal membrane oxygenation (ECMO).
Methods: Between July 2016 and October 2017, 6 consecutive complex and high-risk coronary disease patients underwent routine ECMO-supported PCI.
Results: The average age of the patients was 70.5±11.98, and half of them (50%) were male. The mean creatinine (Cr) was 188.67±151.68 µmol/L. The average scores for SYNTAX, SYNTAX II, and LVEF pre the procedure was 41.33±12.14, 47.87±9.45 / 31.55±8.82, and 44.40±12.58%, respectively. The mean supporting duration of ECMO was 10.50±7.79 h. Regarding the postoperative complication, one case observed lower limp venous thrombosis and another reported infection at the access site. Two patients (33.3%) died for refractory heart failure during the follow-up course of 17.00±9.51 months, and the average net improvement index (NII) was 28.30±25.11% for this period.
Conclusion: With the support of ECMO, the prognosis of complex high-risk coronary disease has shown to be improved by intervention in our study.
Keywords
Percutaneous coronary intervention, mechanical circulation support, extracorporeal membrane oxygenation, prognosis
Introduction
The percutaneous coronary intervention (PCI) has developed rapidly during the past four decades, which has increasingly become one of the mainstream treatments for coronary atherosclerotic heart disease (CHD) [1-3]. Previous studies have shown that revascularization therapy, both PCI and coronary artery bypass graft (CABG), could reduce all-cause mortality and cardiac death among patients with complex high-risk coronary disease in contrast to conventional medication treatments [4, 5]. Regarding the preference for the revascularization strategy, numerous studies recommend CABG to patients who are under such complex high-risk coronary conditions [6-9]. However, due to intricate and asymmetrical characteristics shared among patients like advanced age, complicated multi-organ dysfunction, or previous thoracotomy, the PCI seems to be the only option for those who are unable to tolerate general anaesthesia or surgical procedure of CABG. Also, with the increasing amount of complexity and the risks associated become greater. To complete the revascularization of complicated coronary lesions effectively has increasingly rendered into an urgent focus for cardiologists than ever before [10, 11]. Fortunately, the venoarterial extracorporeal membrane oxygenation (VA-ECMO), as mechanical circulation support (MCS) devices, for such coronary disease during interventional procedures has been applied in the current study and the paralleled mid-term prognosis has been further figured out.
Methods
I Study Population
Case data of this study were retrospectively collected from July 2016 to October 2017 in China-Japan Friendship Hospital. The inclusion criteria were defined as followings: (i) Coronary angiography showed severe coronary lesions (manifested as multivessel disease, unprotected left main (UPLM) disease, and severe coronary artery calcification); (ii)The patients experienced frequent attacks of angina pectoris; (iii)The patients were either unable to tolerate or refuse CABG due to advanced age, severe heart failure, or another organ dysfunction. The risk assessment was given based on the reference of combined clinical, anatomic, and hemodynamic factors and agreed upon all members of a cardiac clinical research team consisting of a primary cardiologist, an interventional cardiologist, and a cardiothoracic surgeon.
II Data Collection and End-Point Definition
This study collected clinical data draw from both in-hospital and follow-up cases, including general clinical information, intraoperative records, and in-hospital and follow-up major adverse cardiac and cerebrovascular events (MACCEs). MACCEs were defined as: (i) All-cause mortality; (ii) Acute myocardial infarction (AMI); (iii) Stroke; (iv) Further revascularization by either PCI or CABG. Additionally, we attempted to use the average of net improvement index (NII), which described as the net area above the curve during the whole period of follow-up time per month from the basic level of left ventricular ejection fraction (LVEF) pre the procedure, to assess the improvement in cardiac function throughout the follow-up period.
$$ NII=\ {\left(S_a-S_b\right)\ }/{\ t} $$
Sa = The area above the basic line.
Sb = The area below the basic line.
t = Follow-up time (month).
III Establishment and Management of ECMO
The ECMO circuitry and instrumentation (Medtronic, Inc., St. Paul, MN, USA) involved a centrifugal machine (Medtronic-550), customized heparin coating integrated package pipeline (including pump and membrane oxygenator), heparin coating femoral venoarterial cannula, air-oxygen mixer, variable temperature water tank, and MAQUET hemoconcentrators. All the patients were received ECMO via femoral venoarterial catheterization (VA mode). The auxiliary flow rate was set to 40-50 ml/ (kg × min). The arterial puncture catheter was used to monitor the pressure, and the mean arterial pressure (MAP) was maintained at the range of 50-70 mmHg. In the case of the unstable hemodynamic or malignant arrhythmia, the auxiliary flow was increased to 3 L/min to preserve tissue perfusion. An air-oxygen mixer can provide 50-60% concentration of oxygen with the simultaneous demand of ECMO support, with a ventilation-to-blood flow ratio stabilized at 0.6-1:1 and PCO2 maintained at 35-45 mmHg. Activated clotting time (ACT) was maintained at the range of 150-180s and under the unintermittent detection every 2 hours during the operation.
IV Procedure and Protocol for PCI
All the patients received the antiplatelet therapy and unfractionated heparin (UFH) for 100 U/kg intra-arterial injection before starting the procedure. Severe coronary calcified lesions indicated the rotational atherectomy, and the drug-eluting stent (DES) was placed at the severe stenosis lesions. The successful revascularization of the target lesion was defined as the residual stenosis < 20% and with thrombolysis in myocardial infarction (TIMI) grade 3 flow.
V Weaning of ECMO
After the PCI, the ECMO auxiliary flow was reduced gradually until the machine shut down completely if the patient was stable in hemodynamically. Successful weaning is defined as hemodynamic stability for 72 hours after weaning with no needs for additional circulation supporting.
VI Statistical Methods
Descriptive data analyses were observed as mean±SD. Relationships of different types of variables were examined using the t-test and chi-square test in STATA (14.1 version, StataStata College Station, Texas). P-value < 0.05 was considered statistically significant.
Results
I Baseline Characteristics
According to the inclusion criteria, six patients, three males and three females, completed the complex high-risk coronary PCI supported by ECMO. The average age was 70.5±11.98 years old. Five patients (83.33%) reported a history of diabetes, and four of them (66.67%) stated a history of chronic kidney disease (CKD). The average Cr was 188.67±151.68 µmol/L, and the average LVEF was 46.50±13.58% pre the procedure (Table 1).
Table 1: Baseline information of preprocedural.
|
Case number |
Gender |
Age |
Date of procedure |
Hypertension |
DM |
Smoking |
CABG |
Cr (µmol /L) |
|
1 |
M |
67 |
2016.07.11 |
N |
Y |
Y |
Y |
350 |
|
2 |
F |
54 |
2017.04.05 |
N |
Y |
N |
N |
121 |
|
3 |
M |
78 |
2017.05.08 |
Y |
Y |
N |
N |
117 |
|
4 |
F |
60 |
2017.05.11 |
Y |
Y |
N |
N |
411 |
|
5 |
M |
83 |
2017.06.12 |
Y |
Y |
N |
N |
68 |
|
6 |
F |
81 |
2017.10.23 |
N |
N |
N |
N |
65 |
|
Mean/Percentages % |
M=50% |
70.50±11.98 |
|
50.00% |
83.33% |
16.67% |
16.67% |
188.67±151.68 |
*DM: diabetes mellitus; CABG: coronary artery bypass grafting; Cr: creatinine; Y: Yes; N: No.
All the patients had triple vessel disease, and even among 4 of them had UPLM disease. The average SYNTAX score was 41.33 ± 12.14, and the SYNTAX II score was 47.87±9.45 / 31.55±8.82 (Table 2).
Table 2: Coronary lesion score and interventional treatment and follow-up.
|
Case number |
1 |
2 |
3 |
4 |
5 |
6 |
Percentage / Mean±SD |
|
|
Left main disease |
Y |
Y |
N |
N |
Y |
Y |
66.70% |
|
|
Triple vessel disease |
Y |
Y |
Y |
Y |
Y |
Y |
100% |
|
|
Bifurcation lesions |
Y |
Y |
Y |
N |
N |
Y |
66.70% |
|
|
Severe calcification |
Y |
N |
Y |
Y |
Y |
Y |
83.33% |
|
|
Gensini Sore |
312 |
126 |
64 |
189 |
96 |
155 |
157.00±87.62 |
|
|
SYNTAX Score |
59 |
49 |
27 |
29 |
44 |
40 |
41.33±12.14 |
|
|
SYNTAX II Score |
58.0/38.4 |
57.4/22.2 |
38.9/37.9 |
50.5/22.7 |
35.0/41.9 |
47.4/26.2 |
47.87±9.50 / 31.55±8.82 |
|
|
Coronary of atherectomy |
LAD, LCX |
N |
LAD; LCX |
LAD |
LAD |
LM-LAD, RCA |
8 in total |
|
|
Number of stents |
3 |
2 |
3 |
2 |
2 |
3 |
2.50±0.55 |
|
|
CR |
Y |
Y |
Y |
Y |
Y |
Y |
100% |
|
|
Support time (h) |
9 |
26 |
6 |
10 |
6 |
6 |
10.50±7.79 |
|
|
Arrhythmia |
ST |
N |
N |
N |
N |
N |
16.67% |
|
|
CRRT |
N |
Y |
N |
Y |
N |
N |
33.33% |
|
|
LVEF (%) |
Basic |
35 |
30 |
63 |
40 |
50 |
39 |
42.83±11.89 |
|
Mean NII |
0.00 |
45.32 |
2.20 |
46.88 |
16.95 |
58.43 |
28.30±25.11 |
|
|
Follow-up time (months) |
N |
25 |
25 |
4 |
10 |
21 |
20.25±7.09 |
|
|
Complication |
N |
infection |
N |
N |
thrombus |
N |
33.33% |
|
|
MACCEs |
Died |
Survival |
Survival |
Died |
Survival |
Survival |
33.33% |
|
*CR: complete revascularization , complete revascularization; ST: sinus bradycardia, sinus bradycardia; CRRT: continuous renal replacement; LAD: left anterior descending branch; LCX: left circumflex; RCA: right coronary artery; LM: left main therapy, continuous renal replacement therapy; LVEF: left ventricular ejection fraction; NII: net improvement index; MACCEs: major adverse cardiac and cerebrovascular events; Y: Yes; N: No.
II Result of the Procedures and Prognosis
All the patients completed PCI supported by ECMO successfully. The mean course of ECMO support time was 10.50±7.79 hours, and the average number of DES placed for each patient was 2.5. Although no malignant arrhythmia or other serious complications such as haemorrhage including gastrointestinal and intracranial haemorrhage or acute renal dysfunction reported during the procedure, one case witnessed a catheter-punctured site infection after the procedure, and another case showed deep venous thrombosis in the catheter-punctured site.
Figure 1: LVEF changes during follow-up and fitting curve.
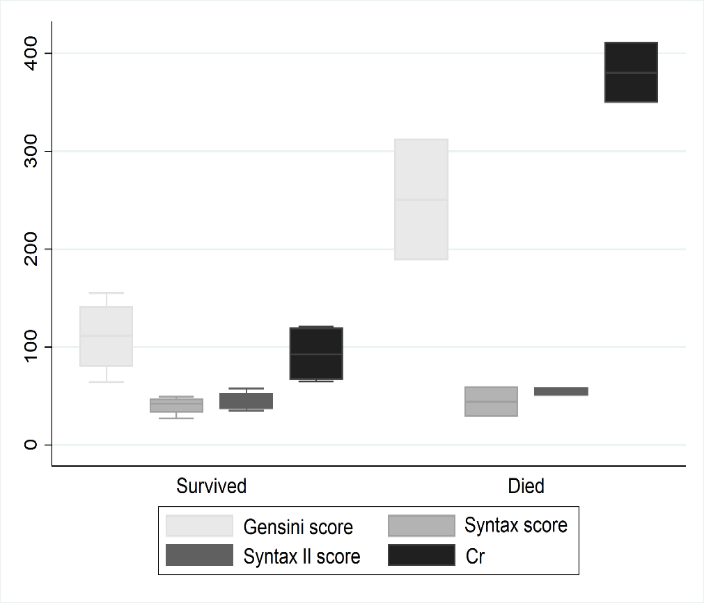
All ECMO were weaned successfully, and no hospitalized deaths occurred. The survival rate throughout the follow-up period of 17.00±9.51 months was 66.7% (n=4) with 2 patients died for refractory heart failure. The average NII was 28.30±25.11% for the follow-up, and the max NII was 58.43% reported by one case (Table 2, Figure 1). The pre-procedure Gensini score and creatinine (Cr) level were higher in the survived group though this difference was not statistically significant (P = 0.06) (Table 3, Figure 2).
Figure 2: Different prognostic group lesion scores and creatinine comparison.
Table 3: Different prognostic group lesion scores and creatinine comparison.
|
Group |
Gensini score |
Syntax score |
Syntax II score |
Cr (µmol /L) |
|
Survival group |
110.38±39.19 |
40.00±9.42 |
44.58±9.90/32.05±9.35 |
92.75±30.38 |
|
Death group |
250.50±86.97 |
44.00±21.21 |
54.25±5.30/30.55±11.10 |
380.50±43.13 |
|
p value |
0.06 |
00.643 |
0.165 |
0.06 |
*Cr: creatinine.
Discussion
The strategy of revascularization for complex high-risk coronary disease had been no more limited to CABG owning to the rapid advancement of the interventional technique and equipment. However, when complicated with severe cardiac insufficiency or multi-organ dysfunction, the patients are prone to circulatory collapse during the interventional procedure. By undergoing the PCI in such a complex high-risk coronary lesion, the guidelines have recommended the MCS devices to keep the hemodynamics stable [12].
The idea of potential and contingent values from the intra-aortic balloon pump (IABP) in providing hemodynamic supports all along the interventional procedure of complex high-risk coronary disease seems plausible and convincing among the cardiologists during the past years [13]. An increasing number of new studies, however, have started to challenge the entrenched viewpoint and argue the opposite that IABP contributes little to the delineated situation [14, 15]. Moreover, other options, like percutaneous left ventricular assist devices (PVADs) such as Impella and TandemHeart, have been demonstrated to offer effective yet partial hemodynamic supports for complex coronary lesions [16, 17].
Nevertheless, Becher found that there was no significant difference in MACCEs between the groups that applied the PVADs and not in terms of circulatory assistance in PCIs among 61 cases which had triple vessel disease and severe heart failure [18]. Besides, implied by a meta-analysis designed for MCS in PCI for high-risk coronary disease, although the Impella could provide relatively stable circulatory supports, the mortality between the group using IABP and the another applied the Impella yields no significant difference, and the cost-benefit analysis even suggested a higher cost for Impella 2.5 [19]. Moreover, Kovacic et al. compared the supporting competency between Impella 2.5 and TandemHeart for 68 high-risk coronary disease patients who received the PCI [20]. They found that both MCS could provide stable circulatory assistance but without a major difference in the complication of insertion position or MACEs during 30 days of follow-up. Furthermore, a meta-analysis showed that there was no significant difference in the mortality between IABP and PVADs as the MCS during the PCI procedure for severe heart dysfunction patients, but the relative risk in the PVADs group was higher than that of IABP group [21].
Unlike the PVADs, the ECMO has been applied in maintaining organ perfusion by the artificial heart pump in the replacement of the patient self-heart pump efficiently in interventional procedures over the recent years [22, 13]. In the early 1990s, Teirstein et al. found the optimal time for ECMO insertion [23]. In their study, there were 389 patients in the prophylactic support group and 180 in the standby support group, and both groups achieved comparable success and major rates of complication. Patients with severe left ventricular dysfunction might benefit from the institution of prophylactic ECMO support. Therefore, in the current study, we adapted that strategy in the opportunity which is the institution of prophylactic ECMO support in the interventional procedure.
However, previous discussions about complex high-risk PCI under the assistance of the MCS devices still bog on the nascent stage. To date, there have been few significant results reported on large-scale multi-center and long-term follow-up studies, in particular, focusing on the ECMO supported PCI for complex high-risk coronary disease. A study from Tomasello presented a single-center experience in ECMO supported complex high-risk PCI among 12 patients, with a mean age of 63.5±8.7 years and a mean SYNTAX score of 30.1±10.1. Whereas all patients included in that study survived during a period of a 6-month follow-up though two of them needed further revascularization [24]. As for the mid-term follow-up, Shaukat’s study enrolled five male patients who successfully received the ECMO supported PCI. The mean age for patients was 66.8±8.6 years, and mean LVEF was 26.6±18.0%. The same study further illustrated that ECMO supported PCI were well-tailored for patients with lower LVEF, and there was no MACCE occurrence during the subsequent course of a one-year follow-up [25].
The patients included in our study were observed in a much more serious status, featuring higher age, more complex coronary lesions followed with higher SYNTAX score, UPLM or multivessel disease with heavy calcification, and more severe organ dysfunction such as depressed systolic function or CKD. This noticeable stack of characteristics has thus given a viable opportunity for the MCS assistance intervention procedure to step in. Between the group of the survived and the dead, no significant difference in the Gensini score and the CR level may indicate that the more complicated the situation, the worse the ending. The mean follow-up time in our study is much longer than any of previous ones, achieving 17±9.51 months, and the timespan for the most prolonged case even continued up to 25 months. Besides, we attempted to employ the NII to standardize the LVEF change during the period of follow-up, aiming at exploring the method to eliminate the bias caused by differences from follow-up time. The result showed that the PCIs were all successful, no hospitalized death occurred, and the incidence of MACCEs in the longer term of follow-up was satisfied. According to the previous study, the mortality of medication therapy group represented more than twice compared with that of revascularization group [26]. The same picture was captured in our study. Compared with conservative therapy, the interventional method significantly improved the prognosis of patients facing such complex high-risk situations. Also, based on our follow-up surveys, the LVEF increased post the procedure, strongly confirming the positive significance of revascularization therapy for myocardial perfusion.
Still, ECMO can provide full circulatory support on the one hand but can also introduce complications on the other. With the ECMO supporting time increasing, the number of reported incidences of complications such as haemorrhage, thrombosis, infection, liver or kidney dysfunction, and distal limb ischaemic necrosis climbs gradually [27, 28]. In this study, one case observed catheter-inserted site infection postoperative and another case had venous thrombosis in the catheter-inserted site. For the rest of cases, no other severe complications were observed. Past studies had shown a connection between the mortality and ECMO supporting time, which may due to the increased complications after longer ECMO duration [29]. So, we suggest that ECMO should get weaned off as soon as the patient is hemodynamically stable.
Patients who seek ECMO support are often in a more severe situation, and their complication risk of interventional therapy is thus much higher than those of others. As a result, indications of ECMO supported PCI are advised to go through a stringent evaluation and to be overseen on the case-by-case bases. The current empirical study thus suggests that cardiologists take into account the characteristics of the lesion and related benefits as well as risks when deciding the type of treatment, say intervention or CABG, for individual cases.
This study still bears limitations although it has yielded a good clinical result. First, the sample size is too small to generalize to the rest of the population. Second, due to the absence of a control group, the benefit of ECMO implementation from this observational study needs further investigation.
Conclusion
Our study reveals that VA ECMO can provide effective circulatory support and ensure a great degree of safety during the intervention procedure for complex high-risk coronary disease. More importantly, we find that ECMO supported PCI provides a more viable strategy for treating the complex high-risk coronary disease.
Acknowledgments
We thank patient advisers for the information they provided.
Conflicts of Interest
None.
Consent
All the patients signed informed consent before the initiation of the procedure.
Article Info
Article Type
Research ArticlePublication history
Received: Wed 12, Aug 2020Accepted: Fri 28, Aug 2020
Published: Thu 10, Sep 2020
Copyright
© 2023 Jin-Gang Zheng. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Hosting by Science Repository.DOI: 10.31487/j.JICOA.2020.05.01
Author Info
Zheng Zheng Shuo-Yan An Xiao-Fei Liu Zhe Dong Hu Zhang Zhi-Wei Han Ming-Jian Wang Jin-Gang Zheng
Corresponding Author
Jin-Gang ZhengDepartment of Cardiology, China-Japan Friendship Hospital of Ministry of Health, Beijing, China
Figures & Tables
Table 1: Baseline information of preprocedural.
|
Case number |
Gender |
Age |
Date of procedure |
Hypertension |
DM |
Smoking |
CABG |
Cr (µmol /L) |
|
1 |
M |
67 |
2016.07.11 |
N |
Y |
Y |
Y |
350 |
|
2 |
F |
54 |
2017.04.05 |
N |
Y |
N |
N |
121 |
|
3 |
M |
78 |
2017.05.08 |
Y |
Y |
N |
N |
117 |
|
4 |
F |
60 |
2017.05.11 |
Y |
Y |
N |
N |
411 |
|
5 |
M |
83 |
2017.06.12 |
Y |
Y |
N |
N |
68 |
|
6 |
F |
81 |
2017.10.23 |
N |
N |
N |
N |
65 |
|
Mean/Percentages % |
M=50% |
70.50±11.98 |
|
50.00% |
83.33% |
16.67% |
16.67% |
188.67±151.68 |
*DM: diabetes mellitus; CABG: coronary artery bypass grafting; Cr: creatinine; Y: Yes; N: No.
Table 2: Coronary lesion score and interventional treatment and follow-up.
|
Case number |
1 |
2 |
3 |
4 |
5 |
6 |
Percentage / Mean±SD |
|
|
Left main disease |
Y |
Y |
N |
N |
Y |
Y |
66.70% |
|
|
Triple vessel disease |
Y |
Y |
Y |
Y |
Y |
Y |
100% |
|
|
Bifurcation lesions |
Y |
Y |
Y |
N |
N |
Y |
66.70% |
|
|
Severe calcification |
Y |
N |
Y |
Y |
Y |
Y |
83.33% |
|
|
Gensini Sore |
312 |
126 |
64 |
189 |
96 |
155 |
157.00±87.62 |
|
|
SYNTAX Score |
59 |
49 |
27 |
29 |
44 |
40 |
41.33±12.14 |
|
|
SYNTAX II Score |
58.0/38.4 |
57.4/22.2 |
38.9/37.9 |
50.5/22.7 |
35.0/41.9 |
47.4/26.2 |
47.87±9.50 / 31.55±8.82 |
|
|
Coronary of atherectomy |
LAD, LCX |
N |
LAD; LCX |
LAD |
LAD |
LM-LAD, RCA |
8 in total |
|
|
Number of stents |
3 |
2 |
3 |
2 |
2 |
3 |
2.50±0.55 |
|
|
CR |
Y |
Y |
Y |
Y |
Y |
Y |
100% |
|
|
Support time (h) |
9 |
26 |
6 |
10 |
6 |
6 |
10.50±7.79 |
|
|
Arrhythmia |
ST |
N |
N |
N |
N |
N |
16.67% |
|
|
CRRT |
N |
Y |
N |
Y |
N |
N |
33.33% |
|
|
LVEF (%) |
Basic |
35 |
30 |
63 |
40 |
50 |
39 |
42.83±11.89 |
|
Mean NII |
0.00 |
45.32 |
2.20 |
46.88 |
16.95 |
58.43 |
28.30±25.11 |
|
|
Follow-up time (months) |
N |
25 |
25 |
4 |
10 |
21 |
20.25±7.09 |
|
|
Complication |
N |
infection |
N |
N |
thrombus |
N |
33.33% |
|
|
MACCEs |
Died |
Survival |
Survival |
Died |
Survival |
Survival |
33.33% |
|
*CR: complete revascularization , complete revascularization; ST: sinus bradycardia, sinus bradycardia; CRRT: continuous renal replacement; LAD: left anterior descending branch; LCX: left circumflex; RCA: right coronary artery; LM: left main therapy, continuous renal replacement therapy; LVEF: left ventricular ejection fraction; NII: net improvement index; MACCEs: major adverse cardiac and cerebrovascular events; Y: Yes; N: No.
Table 3: Different prognostic group lesion scores and creatinine comparison.
|
Group |
Gensini score |
Syntax score |
Syntax II score |
Cr (µmol /L) |
|
Survival group |
110.38±39.19 |
40.00±9.42 |
44.58±9.90/32.05±9.35 |
92.75±30.38 |
|
Death group |
250.50±86.97 |
44.00±21.21 |
54.25±5.30/30.55±11.10 |
380.50±43.13 |
|
p value |
0.06 |
00.643 |
0.165 |
0.06 |
*Cr: creatinine.

References
- Ali Pourdjabbar, Lawrence Ang, Ryan R Reeves, Mitul P Patel, Ehtisham Mahmud (2017) The Development of Robotic Technology in Cardiac and Vascular Interventions. Rambam Maimonides Med J 8: e0030. [Crossref]
- Christina Tan, Richard A Schatz (2016) The History of Coronary Stenting. Interv Cardiol Clin 5: 271-280. [Crossref]
- Giuseppe Gargiulo, Marco Valgimigli, Davide Capodanno, John A Bittl (2017) State of the art: duration of dual antiplatelet therapy after percutaneous coronary intervention and coronary stent implantation - past, present and future perspectives. EuroIntervention 13: 717-733. [Crossref]
- Yong Liu, Yuanhui Liu, Hualong Li, Yingling Zhou, Wei Guo et al. (2016) Percutaneous coronary intervention for chronic total occlusion improved prognosis in patients with renal insufficiency at high risk of contrast-induced nephropathy. Sci Rep 6: 21426. [Crossref]
- Jing Jing Xu, Yin Zhang, Lin Jiang, Jian Tian, Lei Song et al. (2018) Comparison of Long-term Outcomes in Patients with Premature Triple-vessel Coronary Disease Undergoing Three Different Treatment Strategies: A Prospective Cohort Study. Chin Med J 131: 1-9. [Crossref]
- Waleed E Ali, Satyanarayana R Vaidya, Sylvester U Ejeh, Kingsley U Okoroafor (2018) Meta-analysis study comparing percutaneous coronary intervention/drug eluting stent versus coronary artery bypass surgery of unprotected left main coronary artery disease: Clinical outcomes during short-term versus long-term (> 1 year) follow-up. Medicine 97: e9909. [Crossref]
- Xin Lin Zhang, Qing Qing Zhu, Jing Jing Yang, Yu Han Chen, Yang Li et al. (2017) Percutaneous intervention versus coronary artery bypass graft surgery in left main coronary artery stenosis: a systematic review and meta-analysis. BMC Med 15: 84. [Crossref]
- Glenn N Levine, Eric R Bates, James C Blankenship, Steven R Bailey, John A Bittl et al. (2016) 2015 ACC/AHA/SCAI Focused Update on Primary Percutaneous Coronary Intervention for Patients With ST-Elevation Myocardial Infarction: An Update of the 2011 ACCF/AHA/SCAI Guideline for Percutaneous Coronary Intervention and the 2013 ACCF/AHA Guideline for the Management of ST-Elevation Myocardial Infarction. J Am Coll Cardiol 67: 1235-1250. [Crossref]
- Thomas M Bashore, Stephen Balter, Ana Barac, John G Byrne, Jeffrey J Cavendish et al. (2012) 2012 American College of Cardiology Foundation/Society for Cardiovascular Angiography and Interventions expert consensus document on cardiac catheterization laboratory standards update: A report of the American College of Cardiology Foundation Task Force on Expert Consensus documents developed in collaboration with the Society of Thoracic Surgeons and Society for Vascular Medicine. J Am Coll Cardiol 59: 2221-2305. [Crossref]
- Mario Gössl, David P Faxon, Malcolm R Bell, David R Holmes, Bernard J Gersh (2012) Complete versus incomplete revascularization with coronary artery bypass graft or percutaneous intervention in stable coronary artery disease. Circ Cardiovasc Interv 5: 597-604. [Crossref]
- Edward McNulty (2013) Coronary Artery Bypass Grafting Versus Percutaneous Coronary Intervention for Unprotected Left Main Disease - A Review. Interv Cardiol 8: 14-18. [Crossref]
- Glenn N Levine, Eric R Bates, James C Blankenship, Steven R Bailey, John A Bittl et al. (2011) 2011 ACCF/AHA/SCAI Guideline for Percutaneous Coronary Intervention. A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Society for Cardiovascular Angiography and Interventions. Circulation 124: e574-e651. [Crossref]
- Cesar Y Guerrero Miranda, Shelley A Hall (2019) Cardiac catheterization and percutaneous intervention procedures on extracorporeal membrane oxygenation support. Ann Cardiothorac Surg 8: 123-128. [Crossref]
- Bradley W Ternus, Jacob C Jentzer, Abdallah El Sabbagh, Mackram F Eleid, Malcolm R Bell et al. (2017) Percutaneous Mechanical Circulatory Support for Cardiac Disease: Temporal Trends in Use and Complications Between 2009 and 2015. J Invasive Cardiol 29: 309-313. [Crossref]
- Petronio Generoso Thomaz, Leonel Adelino Moura Júnior, Giovana Muramoto, Renato Samy Assad (2017) Intra-aortic balloon pump in cardiogenic shock: state of the art. Rev Col Bras Cir 44: 102-106. [Crossref]
- Aung Myat, Niket Patel, Shana Tehrani, Adrian P Banning, Simon R Redwood et al. (2015) Percutaneous circulatory assist devices for high-risk coronary intervention. JACC Cardiovasc Interv 8: 229-244. [Crossref]
- Nisha A Gilotra, Gerin R Stevens (2015) Temporary mechanical circulatory support: a review of the options, indications, and outcomes. Clin Med Insights Cardiol 8: 75-85. [Crossref]
- Tobias Becher, Frederik Eder, Stefan Baumann, Dirk Loßnitzer, Berit Pollmann et al. (2018) Unprotected versus protected high-risk percutaneous coronary intervention with the Impella 2.5 in patients with multivessel disease and severely reduced left ventricular function. Medicine 97: e12665. [Crossref]
- Health Quality Ontario (2017) Percutaneous Ventricular Assist Devices: A Health Technology Assessment. Ont Health Technol Assess Ser 17: 1-97. [Crossref]
- Jason C Kovacic, Huy T Nguyen, Rucha Karajgikar, Samin K Sharma, Annapoorna S Kini (2013) The Impella Recover 2.5 and TandemHeart ventricular assist devices are safe and associated with equivalent clinical outcomes in patients undergoing high-risk percutaneous coronary intervention. Catheter Cardiovasc Interv 82: E28-E37. [Crossref]
- Fang Bin Hu, Lian Qun Cui (2018) Percutaneous left ventricular assist device vs. intra-aortic balloon pump in patients with severe left ventricular dysfunction undergoing cardiovascular intervention: A meta-analysis. Chronic Dis Transl Med 4: 260-267. [Crossref]
- Georg Trummer, Christoph Benk, Rolf Klemm, Paul Biever, Johannes Kalbhenn et al. (2013) Short-term heart and lung support: extracorporeal membrane oxygenation and extracorporeal life support. Multimed Man Cardiothorac Surg 2013: mmt008. [Crossref]
- P S Teirstein, R A Vogel, G Dorros, S H Stertzer, M G Vandormael et al. (1993) Prophylactic versus standby cardiopulmonary support for high risk percutaneous transluminal coronary angioplasty. J Am Coll Cardiol 21: 590-596. [Crossref]
- Salvatore Davide Tomasello, Marouane Boukhris, Vladimir Ganyukov, Alfredo R Galassi, Dmitri Shukevich et al. (2015) Outcome of extracorporeal membrane oxygenation support for complex high-risk elective percutaneous coronary interventions: A single-center experience. Heart Lung 44: 309-313. [Crossref]
- Arslan Shaukat, Katarzyna Hryniewicz Czeneszew, Benjamin Sun, Karol Mudy, Kelly Wilson et al. (2018) Outcomes of Extracorporeal Membrane Oxygenation Support for Complex High-Risk Elective Percutaneous Coronary Interventions: A Single-Center Experience and Review of the Literature. J Invasive Cardiol 30: 456-460. [Crossref]
- Lin Jiang, Lianjun Xu, Lei Song, Zhan Gao, Jian Tian et al. (2018) Comparison of three treatment strategies for patients with triple-vessel coronary disease and left ventricular dysfunction. J Interv Cardiol 31: 310-318. [Crossref]
- James Thomas, Vadim Kostousov, Jun Teruya (2018) Bleeding and Thrombotic Complications in the Use of Extracorporeal Membrane Oxygenation. Semin Thromb Hemost 44: 20-29. [Crossref]
- Kathleen M Lamb, Hitoshi Hirose (2017) Vascular Complications in Extracoporeal Membrane Oxygenation. Crit Care Clin 33: 813-824. [Crossref]
- Myles Smith, Alexander Vukomanovic, Daniel Brodie, Ravi Thiagarajan, Peter Rycus et al. (2017) Duration of veno-arterial extracorporeal life support (VA ECMO) and outcome: an analysis of the Extracorporeal Life Support Organization (ELSO) registry. Crit Care 21: 45. [Crossref]
